 
Its name is derived from the Greek word thallos, which means twig or green shoot. Nickel oxide Molecular Formula NiO Average mass 74. Thallium has also been used with sulfur, selenium or arsenic to produce low melting glasses which become fluid between 125 and 150 ☌, while thallium oxide has been used to produce glasses with a high index of refraction, and is also used in the manufacture of photo cells. The invention relates to a pulse plating method of an Ag-Ni alloy for an electric contact material. Thallium bromide-iodide crystals have been used as infrared optical materials. 1: Chemical hot packs and cold packs work because of the heats of solution of. For calcium chloride, H soln 82.8 kJ/mol. The molar heat of solution ( H soln) of a substance is the heat absorbed or released when one mole of the substance is dissolved in water. This ability makes the compound useful in photocells. CaCl 2 ( s) Ca 2 + ( a q) + 2 Cl ( a q) + 82.8 kJ. Thallium is primarily used for its electrical conductivity as thallium sulfide, which changes with exposure to infrared light. Thallium is a post-transition metal that is not found free in nature. 
Thallium was discovered by Sir William Crookes in 1861 and first isolated by Claude-Auguste Lamy in 1862. The thallium atom has a radius of 170 pm and a Van der Waals radius of 196 pm. 
The number of electrons in each of thallium's shells is 2, 8, 18, 32, 18, 3 and its electron configuration is 4f 14 5d 10 6s 2 6p 1. Thallium (atomic symbol: Tl, atomic number: 81) is a Block P, Group 13, Period 6 element with an atomic weight of 204.38. How many grams are in 2.34 moles of Sodium hydroxide You will utilize the molar mass you calculated and use dimensional analysis. Thin Film Deposition & Evaporation Materials.Additive Manufacturing & 3D Printing Materials.Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. In this compound, we have one atom of zinc, two atoms of nitrogen (one. For example, take the example of zinc nitrate, or Zn (NO 3) 2. For other compounds, this might get a little bit more complicated. The number of moles per liter in solid AgCl is the same at the start of the reaction as it is when the reaction reaches equilibrium. To complete this calculation, you have to know what substance you are trying to convert. The molar mass will be equal to: (1 atom x 56 grams/mole Fe) + (2 atoms x 35.5 grams/mole of chlorine) 127 grams/mole of iron (II) chloride. The concentration of solid AgCl can be calculated from its density and the molar mass of AgCl. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.Ī common request on this site is to convert grams to moles. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass. 0g of copper (II) oxide 24mg of NaCl (Hint: change to g first) HOMEWORK: Mole Problems 0 Worksheet Finding Molar Mass Using Experimental Data If you do. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. 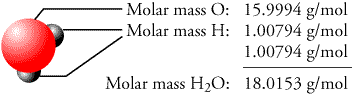
This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. These relative weights computed from the chemical equation are sometimes called equation weights.įinding molar mass starts with units of grams per mole (g/mol). The freezing point of a glucose solution (C6H12O6 molar mass 180.0. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. hydroxide is added to 50.0 ml of 2.5 M nitric acid solution (Assume that there is no. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
